|
Herbal Soaps & Detergents Handbook. Introduction : Soaps and Detergents. Soaps. Soap is any salt of a fatty acid, and usually made by saponification of fatty oil with caustic soda. A specific type of salts lowers the surface tension of water and emulsifies fat having soil particles. But now- a- days herbal soaps are preferred by everyone. Ordinary household bar soap or toilet soap is a mixture of sodium salts of long chain fatty acids. The fatty acids contain 8 to 2. Potassium ammonium and some organic base like triethoanl amine soaps are, like sodium soap, also soluble in water making a specific type of solution called soap solution. Though the definition of soap covers heavy metalic soaps also, these differ from normal soaps in that they are insoluble in water and therefore do not function in the same manner, i. Soaps today are far too sophisticated and are available in a wide variety of forms for specific end uses. The twentieth century has seen transformation of soap manufacture from the conventional batch process of boiling oils with caustic soda in kettles to continuous automated processes involving less steam and also higher recovery of glycerine as a by- product. Similarly processes for conversion of soap base to end products such as washing or toilet soaps have undergone a complete transformation. Laundry soaps were earlier produced as blocks or slabs that were cut into bars and further into cakes. The current process is to spray- cool the molten liquid soap and extrude it as a continuous bar which is cut into billets, stamped and packed. In the case of toilet soaps, the earlier practice was to dry the soap in a shelf drier, followed by milling, storing the milled chips, mixing with other ingredients in a mixer, milling again and extruding. The cut billets were manually stamped and wrapped. At present, toilet soaps are made through continuous processes. Oils or fatty acids and caustic soda are continuously saponified, spray dried, mixed and extruded. Machines are used to stamp, wrap and pack the soap into cardboard boxes. There have been changes in formulation also. The import of tallow or other animal fats is banned in India for use in soaps. Practically all indigenous soaps are made from vegetable oils. Herbal Soaps & Detergents Hand Book. National Institute Of Industrial Re - 536 pages. Herbal based Soaps Shampoos 154197. 154: by special milling method. 159: A Typical Charge. Tallow is completely replaced by different herbal ingredients. Synthetic Detergents. 
The search for substitutes for soaps started during the World Wars because of an acute shortage of oils and fats. Extensive research carried out in Germany and in the U. S. Thus, the formulation of oils for soapmaking has undergone a sea change as practically any oils available in commercial quantities can be suitably treated and used for making laundry as well as toilet soaps. This step is also necessary because the import of oils involves an outflow of foreign exchange which is already in short supply. HERBAL DEODORANT DETERGENT It is a water dispersible emulsion, based on natural essential oils, cleaning, disinfection an deodorisation of kitchen, toilet, living room, office, hospitals, etc. In commercial production, it has. Herbal Soaps & Detergents Handbook PDF View and Downloadable. All rights of this Herbal Soaps & Detergents. Herbal Soaps & Detergents Handbook by H. Panda, ISBN: 8186623590, Rs. We are successfully established supplier and exporter of Herbal Soaps And Detergents Handbook in Delhi, Delhi, India. The book covers Soap Industries Raw Materials, Soaps Manufacture on Tiny Scale, Equipments for Tiny/Cottage. The type of alkali metal used determines the kind of soap product. Sodium soaps, prepared from sodium hydroxide. Manufacturing process of soaps/detergents. The Natural Soapmaking Handbook. Manufacturer of Online Formulation E-Books and Industrial Books - Biscuits, Rusks And Cookies Technology Book, Wheat Flour Mill Technology Book, Packaging Technology And Industries Book and Handbook on Textile Auxiliaries with. Handbook on Herbal Products Medicines. Herbal Soaps & Detergents Handbook. Pages: 10 Size: 59 KB Year: 2013. Soaps, Detergents and Disinfectants Technology Handbook. The term detergent by itself refers specifically to laundry detergent or dish. Herbal Soaps & Detergents Handbook Author: H. Panda Format: Paperback ISBN: 8186623590 Code: NI53 Pages: 536 Price. Mar 4 2012 Soaps Detergents Manufacturing. Linear alkyl benzene is one of the chief raw materials for heavy duty detergents. A number of other heavy duty detergent raw materials have since been marketed each having its own functional benefits. Currently a wide choice of raw materials is available depending on the end use- be it dish washing, for washing ordinary or delicate clothes, or bathing. Earlier, detergents were made by the organised sector by spray- drying the detergent slurry containing the active ingredient, water- softening chemicals, colour, soil suspending chemical, optical brightener, and other builders that improved detergency. The product was sold as powder packed in moisture- proof cartons. Subsequently, detergents were available in different forms such as cakes and liquids for specific end uses. 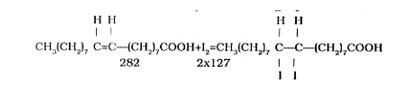
More recently detergents containing enzymes and bleaches are available in the form of powders packed in plastic containers. These detergents are more effective in removing proteinaceous stains from the fabrics and in improving overall detergent efficiency. Physical Properties of Soap. The following properties of pure soap are useful for design engineers and development chemists : Viscosity,Specific heat,Latent heat,Density. The 6. 6% rule,Salt distribution between soap curd and lye,Glycerol distribution between soap curd and lye, and. Rate of drying of soap. Viscosity. When an oil blend is saponified with caustic soda, the resultant soap has a viscosity which is much more than that of either the oil or the caustic soda solution. Above 7. 5o. C the viscosity increase is not significant, but below 7. C it increases rapidly. The viscosity varies in the range of 1. Poise depending on the temperature of the soap and the composition of the oil blend. For the purpose of design of pumps, a viscosity of 1. Poise at 8. 0o. C may be assumed. Specific heat. The specific heat of soaps at 0. Latent heat. The latent heat of fusion of soaps is approximately 3. Density of soap. The density of pure soap is in the range of 0. It may be noted that the figures quoted for viscosity, density, specific heat and latent heat are used frequently. In actual practice, variations around these figures may be expected depending on the composition of soaps. Rule. A soap curd consists of soap hydrate containing 6. Salt distribution between curd and lye. Applying the 6. 6% rule, the free solution in curd contains the same percentage of salt as the spent lye from which the curd has separated. Glycerol distribution between curd and lye. The glycerol distribution in curd and lye is related to the water content of the two layers. Thus,Water in curd. Glycerol in curd. Water in lye. Glycerol in lye. Rate of drying of soap. When soap is allowed to dry by exposure to ambient air, moisture is lost depending on the initial moisture content, its physical structure, duration of exposure, the atmospheric temperature and humidity. Moisture can also be retained or marginally absorbed by the soap when the atmospheric humidity is high. When soap is dried, the moisture that diffuses from the centre to the surface brings with it some soap and electrolytes which eventually appear as 'bloom' especially if the soap contains fillers such as soda ash and sodium silicate. Under certain conditions, soaps such as sodium oleate appear on the surface of the soap as hairy growth. This is a phenomenon observed in the case of soaps processed through spray cooling under vacuum, and derived from natural oils and fats which do not contain rosin. This problem can be eliminated by incorporating 1- 2% rosin in the oil blend, or by using hydrogenated oil instead of natural hard oils as the hard oil component of the oil blend. It is possible that the elaidic acid in hydrogenated oil is responsible for suppressing the hairy growth on soaps. Raw Materials Oils and Fats(The Main Raw Materials for Soaps)Classification of Fats/Oils. Glycerides of various fatty acids solid at room temperature are called fats and those liquid at room temperature are called oils. Thus glycerides heaving higher melting points are termed fats and those having lower melting points are termed oils. And these in turn depend upon the nature of fatty acids content of esters. These acids fall in two series as follows. Saturated series, such as stearic acid. Unsaturated series. Mono enoic acid containing one double bond, such as oleic acid. Poly enoic acids containing more than one double bonds, such as linoleic, linolenic acids. The more unsaturated acids give esters with lower melting points and these are the chief constituents of oils. The more saturated acids containing esters are of higher melting points and are chief constituents of fats (Table- 2). These oils are called fixed oils as distinguished from essential oils and petrolium oils. Fixed oils cannot be distilled without some decomposition under normal atmospheric pressure. Only fixed oil produces soap. Fatty Oils are Further Classified as : Animal origine : These usually occur as fats, such as tallow, lard etc. The liquid types include fish oils, fish liver oils, sperm oils etc. Vegetable origin : These are again sub classified as : A. On the Basis of Degree of Unsaturation. Drying. Iodine value above 1. Tung oil). Semi drying. Iodine value 9. 0- 1. Non drying. Iodine value below 9. B. Enedible Soap stocks (palm oil, coconut oil, mahua oil, rice bran oil other enedible varieties of vegetable oils). C. Edible (sunflower oil, coconut oil, corn oil, soyabean oil, mustard oil, olive oil, cotton- seed oil etc.). Theoretically any oil or fat can be used for making soap. However, in actual practice, the usage of an oil or fat is dictated by the following considerations : Suitability. Availability in commercial quantity. Price. In India import of animal fat is banned. Therefore, the discussion on raw materials will be mainly confined to vegetable oils and fats. Table 2. 1 shows the approximate availability of indigenous oils for soap- making. Other oils used in small quantities are mango kernel, watermelon seed, tobacco seed, khakan, pilu, nahor, undi, dhupa, jute seed, tea- seed etc. The physical hardness of soap depends on the type of oil used. In order to obtain the right hardness it is common to use a blend of oils for soap- making. This subject will be discussed further in Chapter 3. The usage of an oil is also determined by its price and availability. Table 2. 2 shows the analytical characteristics of common soapmaking oils. Table 2. 1 Availability of oils for Soapmaking (1. Oils. Tonnes*Rice bran. Sal seed. 30,0. 00. Acid Oils. 1,0. 0,0. Castor. 60,0. 00. Groundnut RGII4. 0,0. Coconut. 30,0. 00. Linseed. 30,0. 00. Neem. 20,0. 00. Mowrah. Karanja. 20,0. 00. Kusum. 4,0. 00*Average estimate from ISTMA, DGTD. For a better understanding of the quality of fatty raw materials for soaps, it is necessary to explain the significance of the following characteristics : Colour. Saponification value. Iodine value. FFA content. Titre. Fatty acids, containing OH and CO groups. Colour. All oils possess a characteristic colour. Light coloured oils are preferred for making soap. The colour of an oils can be measured quantitatively by using an instrument called tintometer.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2016
Categories |
 RSS Feed
RSS Feed
